Author: Sanudi Sarithma Wilegoda

Air pollution refers to the release of pollutants into the air that affect human health and the health of the whole planet. According to the World Health Organization (WHO), each year, indoor and outdoor air pollution is responsible for nearly seven million deaths worldwide. Urban air pollution is influenced by factors such as population density, energy production, primarily through the burning of fossil fuels, Industrial activities, and vehicle emissions. But thanks to advances in photocatalytic air purification technology, surfaces coated with special materials like Titanium dioxide can break down harmful pollutants to safer and simpler compounds being exposed to light. So, how does this work, and does this give solutions to the air pollution?
Normally, traditional air purifiers (electrostatic smoke precipitator) consume a significant amount of energy and may produce ozone as a harmful byproduct. Mostly, these methods require frequent maintenance with extra operational costs. And also, they are designed for small indoor environments, not suitable for scalable large areas or outdoor use. But photocatalytic oxidation is an efficient technique for removing organic pollutants from the air. Photocatalysts are semiconductors that catalyse reactions when exposed to light. The photoelectrochemical properties of TiO2 are the basis of modern photocatalysis techniques. TiO2 has shown several advantages, including low energy consumption, exceptional efficiency, resistance to acids, ease of production, strong oxidizing ability, chemical stability and insolubility in water. Photocatalysts are involved in accelerating the oxidation process under light irradiation. The process involves interactions between light irradiation and the photocatalyst, generating reactive species that oxidize and break down organic molecules. When protons from the light source strike the photocatalytic surface, electrons within the catalyst react and create electron-hole pairs. These pairs lead to the oxidation process. That holes react with water molecules and produce hydroxyl radicals, while the electrons reduce oxygen molecules to form superoxide anions. Both species accelerate the degradation of pollutants, like organic pollutants.
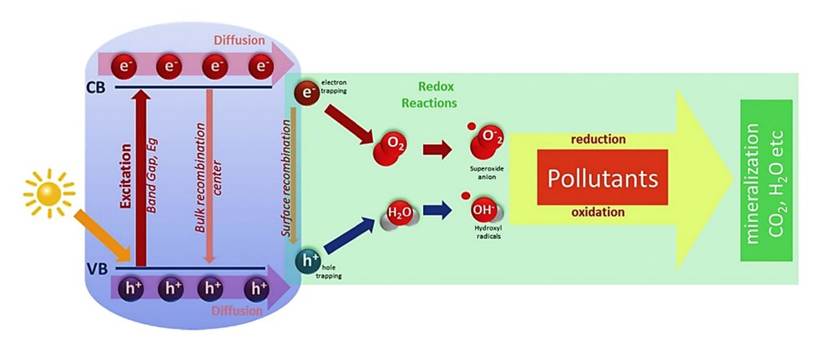
Figure 1; Mechanism of photocatalytic oxidation
As a solution for air pollution, Belgium, in recent years, introduced photocatalytic materials as a construction material (concrete). Under the influence of sunlight, the photoactive TiO2 is activated on the surfaces. Then the pollutants are oxidized due to the presence of photocatalysts and precipitated on the surface of the material. They can be removed from the surfaces by rain or washing with water. Milan and Rome have tested photocatalytic paving bricks and wall paints as a purification material. The pilot project showed up to 40-60% NOx reduction in high traffic zones. Now these photocatalytic materials are used in bridge constructions, graffiti repellent coatings, maintaining water purity, etc. The SARS-CoV-2 (COVID-19) pandemic opened new opportunities for the development of new photocatalytic air treatment units. These photocatalytic systems inactivate the COVID-19 virus, as well as other viruses. Converting hazardous pollutants and volatile organic compounds into safer byproducts reduces air pollution, improves air quality, enhances community welfare, and lowers health risks. Photocatalytic surfaces have benefits such as self-cleaning properties, graffiti resistance, action against microbes, anti-fouling properties, energy savings, sustainability, minimal maintenance, and odor reduction.
The most possible technical limitation of photocatalyst purification surfaces is the low accessibility of gaseous contaminants. In addition, undesirable volatile intermediate compounds can be released. Because of the cost of ingredients, photocatalytic surface materials can be more expensive. The activation of these surfaces always depends on the percentage of light exposure. The photocatalytic surface efficiency can vary based on climate, the amount of pollutant present, direction, and intensity of sunlight.
Future trends of photocatalytic oxidation for the future development of CSP (Concentrated Solar Power) photoreactors. In 2022, visible light-activated photocatalysts, such as graphene oxide modified Ag/Ag2O/BiPO4/Bi2WO6 developed, showed excellent photocatalytic capabilities. Based on the last 5 years of research studies, future development of photocatalytic oxidative mechanisms may focus on the innovation and applications of high-performance immobilized photocatalysts. Those achievements will facilitate high-efficiency photocatalytic reactions and low costs, finally realizing industrial applications.
References:
Chen, F., Zhang, W., Mfarrej, M. F. B., Saleem, M. H., Khan, K. A., Ma, J., Raposo, A., & Han, H. (2024). Breathing in danger: Understanding the multifaceted impact of air pollution on health impacts. Ecotoxicology and Environmental Safety, 280. https://doi.org/10.1016/j.ecoenv.2024.116532
Javed, M. F., Shahab, M. Z., Asif, U., Najeh, T., Aslam, F., Ali, M., & Khan, I. (2024). Evaluation of machine learning models for predicting TiO2 photocatalytic degradation of air contaminants. Scientific Reports, 14(1). https://doi.org/10.1038/s41598-024-64486-7
Title: A brief survey of the practicality of using photocatalysis to purify the ambient air (indoors or outdoors) or air effluents. (2019). https://www.elsevier.com/open-access/userlicense/1.0/
Pichat, P. (2019). A brief survey of the practicality of using photocatalysis to purify the ambient air (indoors or outdoors) or air effluents. Applied Catalysis B: Environmental, 245, 770–776. https://doi.org/10.1016/j.apcatb.2018.12.027
Pham, H. D., Tekalgne, M. A., Do, H. H., Nguyen, V. H., Vo, D. V. N., Cheng, C. K., Shokouhimehr, M., Nguyen, C. C., Singh, P., Raizada, P., Kim, S. Y., & Le, Q. van. (2021). Emerging photocatalysts for air purification. Materials Letters, 288. https://doi.org/10.1016/j.matlet.2021.129355




